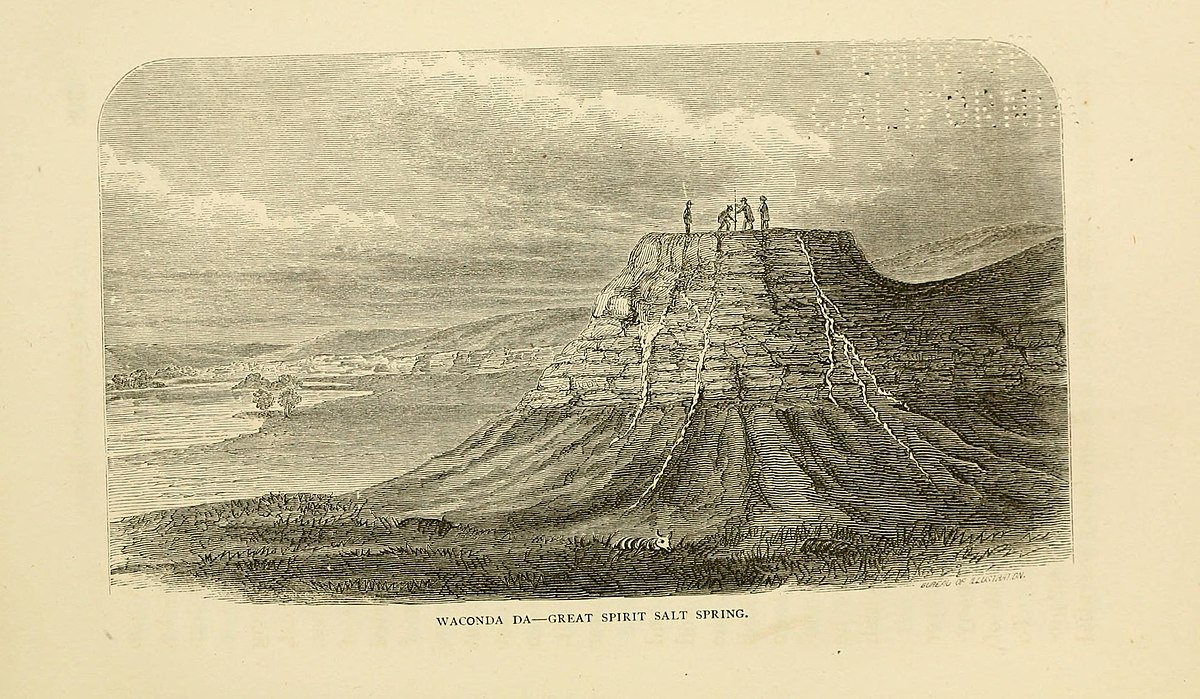
The “Waconda” or Great Spirit Spring, which is situated in Mitchell County, Kansas, about two miles east of Cawker City, has been described in detail by G. E. Patrick in vol. vii, p. 22, Transactions of the Kansas Academy of Science. An analysis of the water, and of the rock forming the mound on which the spring is located, is also given.
The spring is upon a conical, limestone mound 42 feet in height, and 150 feet in diameter at the top. The pool itself is a nearly circular lake about 50 feet in diameter, 35 feet deep, and the water rises to within a few inches of the top of the basin. There is a level space on all sides of the spring so wide that a carriage can be readily driven around it.
There is but little indication of organic matter in the water of the large spring, though there is a slimy white deposit adhering to the bottom and sides, but the water is colorless, clear, and transparent. The excess of water, instead of overflowing the bank, escapes by numerous small fissures, from 10 to 20 feet down on the sides, especially on the side away from the bluff. In these lateral springs there is an abundance of green algæ, and a whitish scum, which seems to be detached from the bottom and to float to the surface. This has a slimy, granular feeling suggesting in a very marked manner hydrated silica.
The mound is situated within about 200 feet of a limestone bluff, which rises perhaps 20 feet above the level of the spring. The natural inference would be that the harder material of the mound protected it from the erosion which carried away the rock in the valley of the Solomon on the south, and the rock between the spring and the bluff.
Is it not possible however that the mound has been really made by the successive deposits from the spring? Although the mound is plainly stratified, this need not interfere with the theory, for the water may have been intermittent in its flow. The rock is very porous, and on being ground to a thin section is shown to be concretionary in structure.
An analysis of the water of the spring (loc. cit.) showed that it contained over 1120 grains of mineral matter per gallon, of which 775 grains were sodium chloride and 206 grains sodium sulphate, with 66 grains of magnesium sulphate, 41 grains of magnesium carbonate, and 31 grains of calcium carbonate. An analysis by the author shows that there are 0.874 grains of silica.
Samples of the rock composing the mound, and of the adjoining bluff were secured, and comparative analyses made, with the following results:
GREAT
COUNTRY SPIRIT
ROCK. MOUND.
Silica and insoluble residue 2.14 4.10
Oxides of Iron and Alumina 3.22 [5]2.66
Sulphuric Anhydride .00 0.34
Carbon Dioxide 40.90 39.10
Calcium Oxide 51.90 49.28
Magnesium Oxide 0.63 1.15
Water and organic matter, undetermined 1.21 [6]3.37
—————— ——————
100.00 100.00
Specific gravity 2.52 2.79
The rocks are entirely different in appearance and structure, that of the mound being twice as hard as that of the bluff. The former contains much organic matter as is shown by blackening when it is heated in a tube and giving off the characteristic odor. The iron is practically of the ferrous variety, probably combined with carbonic acid, and the rock contains traces of chlorides. The particular sample taken was at some distance from the spring, and had been thoroughly exposed to the weather.
The rock of the mound is of just such a character as might have been built up by deposition from the water, as it contains the least soluble constituents of the water. The process of solidification would have been assisted by the silica in the water, forming insoluble cementing silicates, as noticed by Prof. Patrick. The analysis given above shows that there is abundant silica in the water for this purpose.
Mention has been made of the organic growth in the adjacent springs. The mixed scum on being heated changes from a dull green to a vivid grass green, and if ignited it swells up and emits an ill-smelling vapor, which is evidently nitrogenous in its character. A grayish white ash is left, which contains much carbonate of lime. This is evidently freshly deposited, as it is entangled in the algæ in granular lumps.
A specimen of the white scum, noticed above, only slightly mixed with the green algæ, was analyzed. The acid solution of the ash contains 1.26 percent of soluble silica. This was of course a combined silica, probably calcium silicate, which becomes the cementing material in the rock. In another sample of ash, after removing all the substances soluble in hot water, the residue was found to contain 76.46 per cent of silica.
The siliceous residue from the scum was examined by Dr. S. W. Williston. It consists mostly of diatoms. He recognized
Navicula— 2 species
Nitzschia— 2 species
Asteronella— 1 species.
All three genera are found both in fresh and salt or brackish water.
The green material consists essentially of Oscillaria and Confervæ. If the scum is allowed to stand for a short time a very strong sulphuretted odor is developed, strangely suggestive of saltwater marshes or mud flats; and indeed the same odor is noticed in the vicinity of the spring. No characteristic saltwater organisms, that should occasion this peculiar odor have, however, yet been observed here. A more extended and special study of the organic life of these interior saltwater marshes and springs would be of great interest.